Pharma Manufacturing
Advanced Manufacturing Excellence
At Le-Vanza Food & Herbals , our infrastructure reflects our commitment to quality, safety, and operational excellence. We operate with a modern and well-equipped manufacturing facility designed to meet stringent pharmaceutical industry standards.
Our WHO-GMP compliant infrastructure is built to ensure precision, hygiene, and efficiency at every stage of production. From raw material handling to final product packaging, every process is carried out under strict quality control and regulatory compliance.
Equipped with advanced machinery and updated technology, our production units are designed for high efficiency while maintaining consistent product quality. Dedicated quality control laboratories conduct rigorous testing and validation to ensure that every batch meets defined safety and efficacy standards.
Modern Manufacturing Facility
Our advanced manufacturing infrastructure is designed to ensure precision, safety,and efficiency across all pharmaceutical production processes.

WHO-GMP Certified
Manufacturing areas designed and operated in compliance with GMP guidelines to ensure consistent quality and safety.

Advanced Machinery
Equipped with modern and automated equipment to achieve accuracy, efficiency, and high production standards.

Controlled Environment
Strict temperature, humidity, and hygiene controls to maintain product integrity throughout production.

In-Process Monitoring
Continuous monitoring and quality checks at every stage to ensure compliance and product consistency.
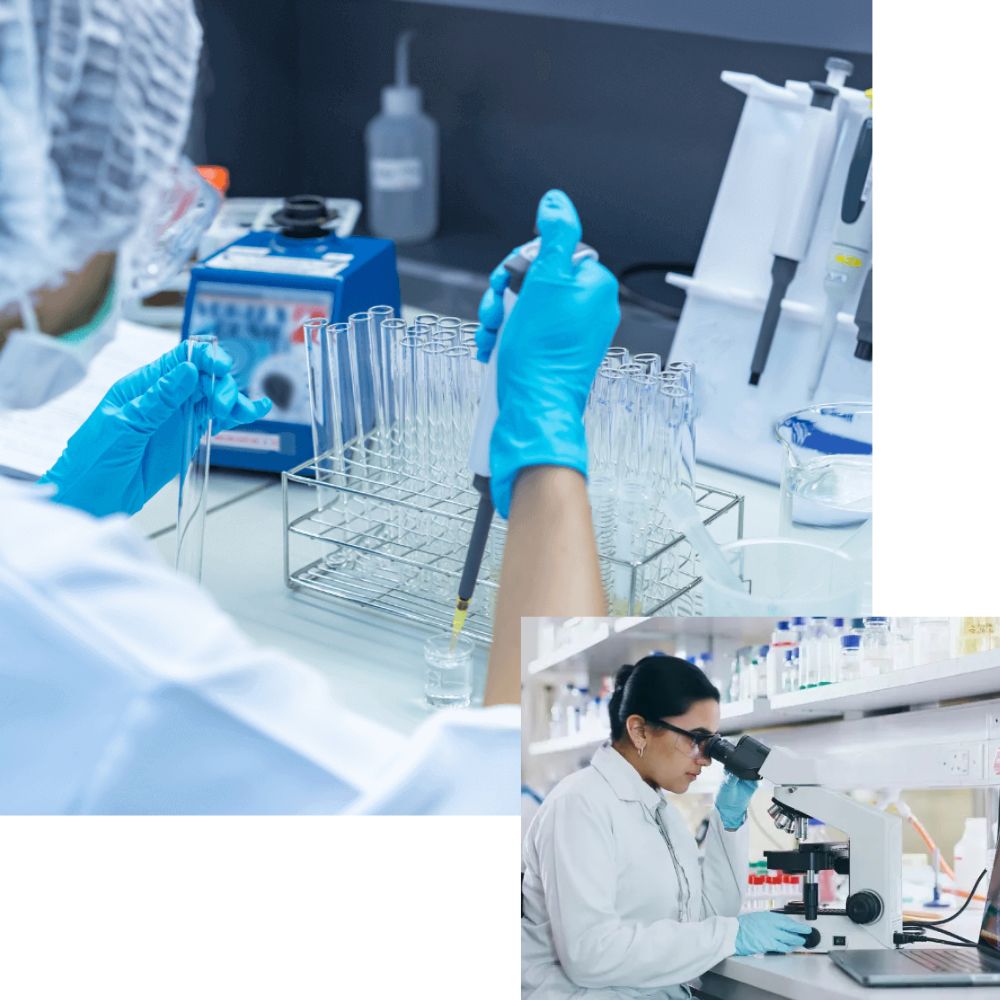
Quality Control & Testing Laboratories
Quality is embedded into every layer of our infrastructure. Our in-house quality control laboratories ensure that every batch meets international pharmaceutical standards before release
Raw Material Testing
Incoming materials are tested for identity, purity, and quality compliance.

In-Process Checks
Quality checks are performed during production to ensure consistency.

Laboratory Analysis
Analytical and microbiological tests confirm safety and stability.

Finished Product Testing
Final products are evaluated against approved quality standards.

Documentation & Release
Results are reviewed, documented, and approved for batch release.
Advanced Infrastructure for Scalable Growth

WHO-GMP Certified

24/7 Quality Monitoring

Advanced Manufacturing Technology

Scalable Production Capacity
Safety, Compliance & Hygiene Standards
Our infrastructure is built to maintain the highest standards of safety, cleanliness, and regulatory compliance. Every stage of our operations—from facility design to daily production processes—is carefully planned to meet strict pharmaceutical guidelines and ensure a controlled manufacturing environment.
WHO-GMP compliant manufacturing systems
Well-defined workflow with proper area zoning
Advanced environmental monitoring and control systems
Routine internal and external audits for quality assurance
Skilled and professionally trained technical team
Full adherence to Schedule M regulatory requirements

